Saturday, August 15, 2020
Ankle Replacement Pictures
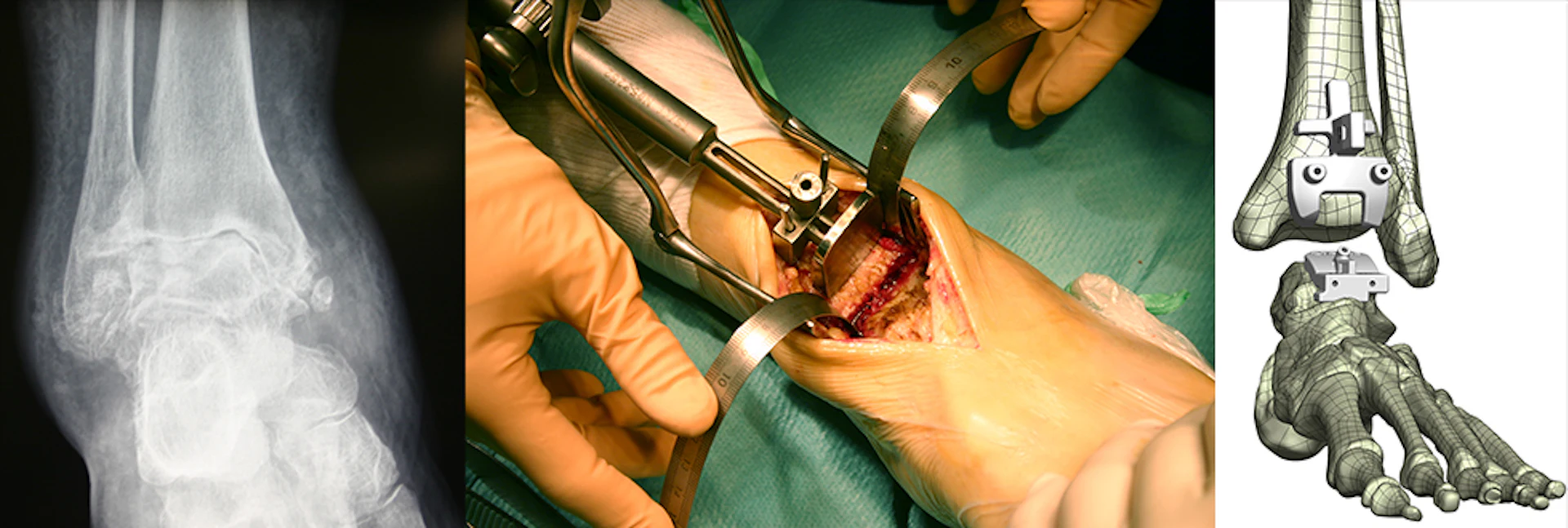
There are many ankle replacement implants on the market. How do you choose the best implant for you? It's really hard to find useful and reliable information on the Internet about the different types of total ankle replacement. In this article we discuss some of the different ankle replacement pictures, implants and prostheses on the market to help you with your ankle replacement recovery.
THE INFINITY ANKLE REPLACEMENT

The Infinity™ ankle replacement is the commonest used implant globally and the commonest ankle replacement in the UK. The implant was launched by Wright Medical in 2014 but in 2020 Wright Medical was acquired by Stryker Inc and so Stryker now own the implant. The Infinity ankle replacement is a two component fixed bearing prosthesis made of titanium or cobalt chrome with a porous titanium plasma spray coating. There is quite a large number of publications to date but the largest outcome series is a multicentre, non-inventor, prospective observational study of 504 implants. All patients had improvement in clinical scores at 6 months, maintained up to 2 years, and the most recently published paper suggests close to 99% of implants had survived 5 years which is very encouraging.
The Infinity™ implant can be used with standard instruments or using a technique known as Patient Specific Instrumentation (PSI) also known as Prophecy™ which is customised to the patients anatomy based on a CT scan performed prior to the surgery.

The Prophecy Infinity Ankle Replacement
THE CADENCE ANKLE REPLACEMENT

The Cadence™ ankle replacement is distributed by Integra LifeSciences. It was launched in 2016 and is a two part fixed bearing prosthesis made of titanium alloy and cobalt chrome alloy. There is limited published outcome data outside of the 2 year surgeon designer series.
THE HINTEGRA (H2/H3) ANKLE REPLACEMENT

The Hintermann Series Total Ankle Replacements began life as the Hintegra distributed by Integra Lifesciences with the first implant in 2000. In 2016 ownership moved to DTMedtech LLC who received FDA premarket approval for a two component version, entitled the H2™. DT Medtech LLC then used 18 years of non US data of their three component design to obtain FDA premarket approval of the H3 prosthesis which was granted in June 2019. The older implant has a lot of published data the largest series of which analysed 722 implants at a mean follow up of 6 years. The data needs to be carefully interpreted, however, since the Indications for Use (IFU) differs for the H2™ and H3™ depending on whether it is sold in the USA or outside of the USA.
THE STAR ANKLE REPLACEMENT

The STAR™ ankle stands for Scandinavian Total Ankle Replacement. It was developed by Hakon Kofoed in Denmark and first implanted in 1981. Since that time there have been more than 5 different versions of the STAR prosthesis. The first four were used in Europe whereas the fifth variety was introduced into the US market in 1998 and FDA approved in 2009.
Although the first STAR™ ankle was a 2 component cemented design and was implanted between 1981–1986, Kofoed then developed the 3 component mobile bearing implant to replace this from 1986–2012. In 1989 the 3rd generation 3 piece design but with HA coating was introduced. Kofoed published improved results from the 3rd generation implant with 12 year survival rates.
In 1998, the base coating of the STAR implant was changed to a rough Titanium plasma so that it could be used in the USA. This is the design used on the US clinical trials for FDA approval (Mann et al., 2011) and is the current design used in the USA & Canada. This is different than the current design used outside of the USA and Canada, as of 1999, which has Calcium Phosphate on top of the Titanium plasma spray (referred to as double coat). Less than 10% of ankle replacements sold in the UK are of the STAR type.
THE VANTAGE ANKLE REPLACEMENT

The Vantage™ ankle replacement is manufactured by Exactech Inc. It was launched in 2017 and is a two part fixed bearing prosthesis in the USA, but a three component mobile bearing prosthesis outside of the USA. It is made of Stainless steel Nitronic 60 and the tibial component has a vertical cage pressfit for initial fixation and peripheral pegs for rotational stability. There is limited published outcome data yet.
Mobile Vs Fixed Bearing Ankle Replacements
Ankle replacements can be two component, where the plastic liner is joined to the tibial implant, known as fixed bearing, or three component, where the plastic liner is mobile and free to move between the implants. Although there are many theoretical advantages of a three component, mobile bearing implant, the evidence from long term use, has suggested that fixed bearing implants outperform mobile bearing implants. This is from National Joint Registry Data, large prospective studies, and randomised controlled trials.
Further Reading
This article is an excerpt of a text book, the Atlas of Ankle Replacements, which catalogues all ankle replacement implants that includes all current implants on the market and also all historic ankle replacement implants. The Editors of The Atlas of Ankle Replacements are Andrew Goldberg OBE and Mr Paul Cooke.
Did you enjoy this article? You may also like; best ankle replacement surgeons
Ankle replacement vs ankle fusion
Jane’s story – the arthritis fix that finally took away my agony